Run Michigan State’s 40‑cow BLV test on a 1,200‑cow herd at 45% prevalence and the math lands on $152,820–$205,200 a year — quietly, in milk, early culls, and condemned carcasses.
Executive Summary: Run Michigan State’s 40‑cow BLV profile on a 1,200‑cow herd at 45% prevalence and the annual loss lands at $152,820–$205,200 — roughly $283–$380 per infected cow in lost milk, early culls, and condemned carcasses. BLV now sits in 88.6–94.2% of U.S. dairy herds, and MSU’s 2015–2016 study of 103 herds across 11 states pegs animal‑level prevalence at 46.5%, up from 40.8% in 1996. Bartlett et al. (J. Dairy Sci. 96:1591–1597, 2013) found ELISA‑positive cows are about 23% more likely to leave the herd, and every 10‑point jump in prevalence costs roughly 209 lb of milk per cow per year. The real leverage isn’t blanket culling — in one herd, 11% of positives drove 64% of new infections, and in another, two cows carried 26% of the total proviral load. Fix the usual leaks (single‑use needles and sleeves, pasteurized or frozen colostrum, qPCR on older positives to tag the top 10–15% super‑spreaders) and a realistic 25–30% recovery is worth about $38,000–$62,000 a year on that same 1,200‑cow herd. If your 40‑cow profile comes back at or above 30–40%, BLV belongs on your P&L as a structural cost, not a footnote. The full piece walks through the Tax Table, the 30/90/365‑day playbook, and where this lands in The Silent Tax series.

Most of the data here comes from Michigan State University’s BLV research program (Bartlett, Erskine, Norby, Coussens, Kendrick, Durst and colleagues) and Dr. Tasia Kendrick’s March 2026 interview on The Dairy Health Blackbelt Podcast (Episode 38). No individual dairies are profiled by name; the “typical 1,200‑cow herd” example is a representative case built from published BLV research and MSU field descriptions. Cross‑reference any numbers here with your own herd’s test results and your vet’s recommendation before acting.
Picture the kind of 1,200‑cow Southeast freestall the Michigan State University BLV team has worked with repeatedly: solid rolling herd average, decent components, nothing on the monthly reports that screams crisis. According to MSU’s BLV research program, many of these herds don’t engage on BLV until their vet pushes for a baseline test, usually because the same older cows keep showing up on hospital lists.

When one of those herds runs MSU’s 40‑cow bovine leukemia virus (BLV) profile and comes back at roughly 45% infected, the cost range the BLV research points to — $283–$380 per infected cow per year — translates to about $152,820–$205,200 a year in lost milk, early culls, and condemned carcasses on a 1,200‑cow herd. That’s not a prediction. It’s money those herds are already paying out, quietly, every year.
“BLV is often present long before it becomes an issue. So if you’re not looking for it on the farm, chances are it’s there.” — Dr. Tasia Kendrick, Associate Professor, MSU Department of Animal Science, on The Dairy Health Blackbelt Podcast (Episode 38, March 2026)
The Silent Margin Leak in 9 Out of 10 U.S. Herds
For years, BLV lived in the “slaughterhouse surprise” category — the disease you only thought about when a carcass came back condemned for lymphoma. That story doesn’t fit anymore.
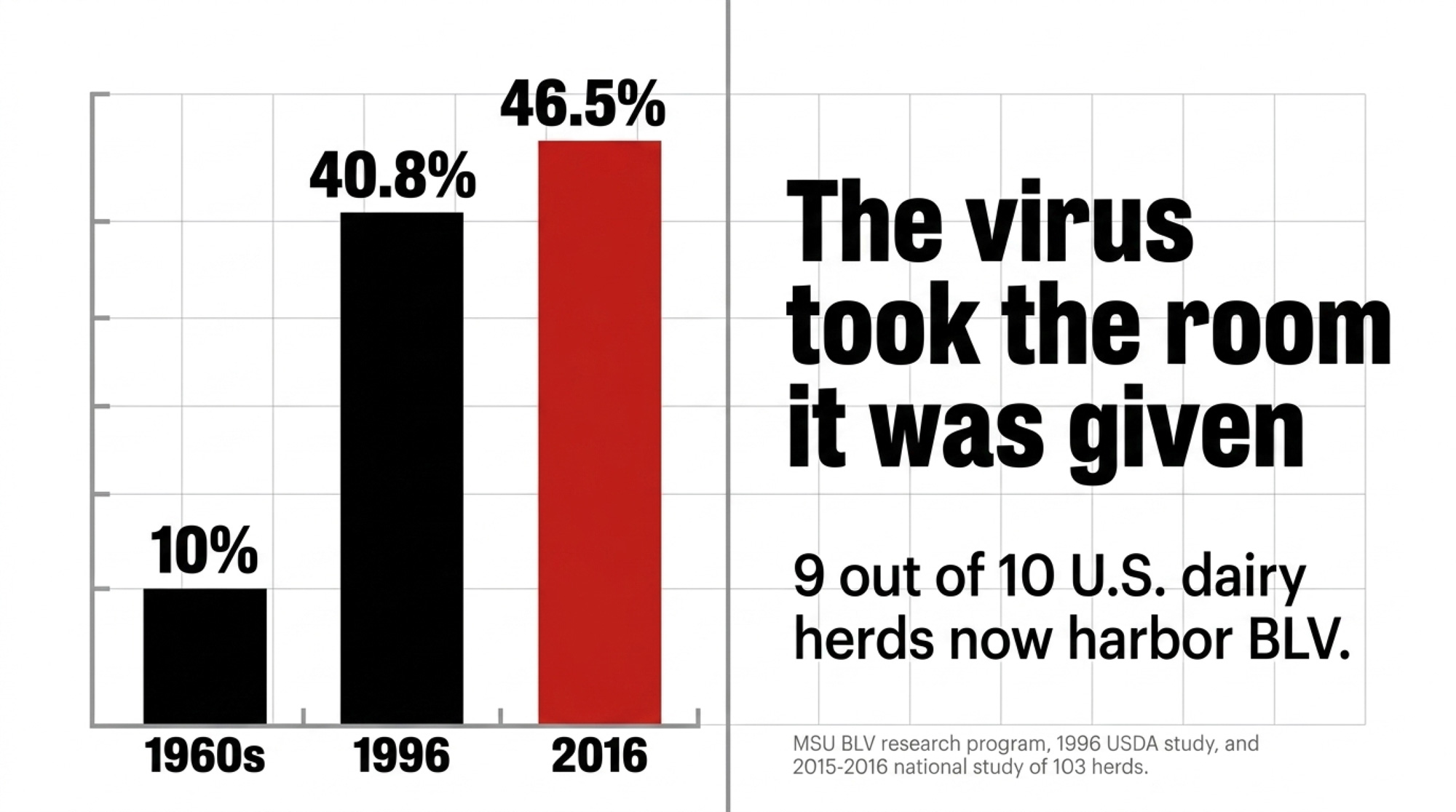
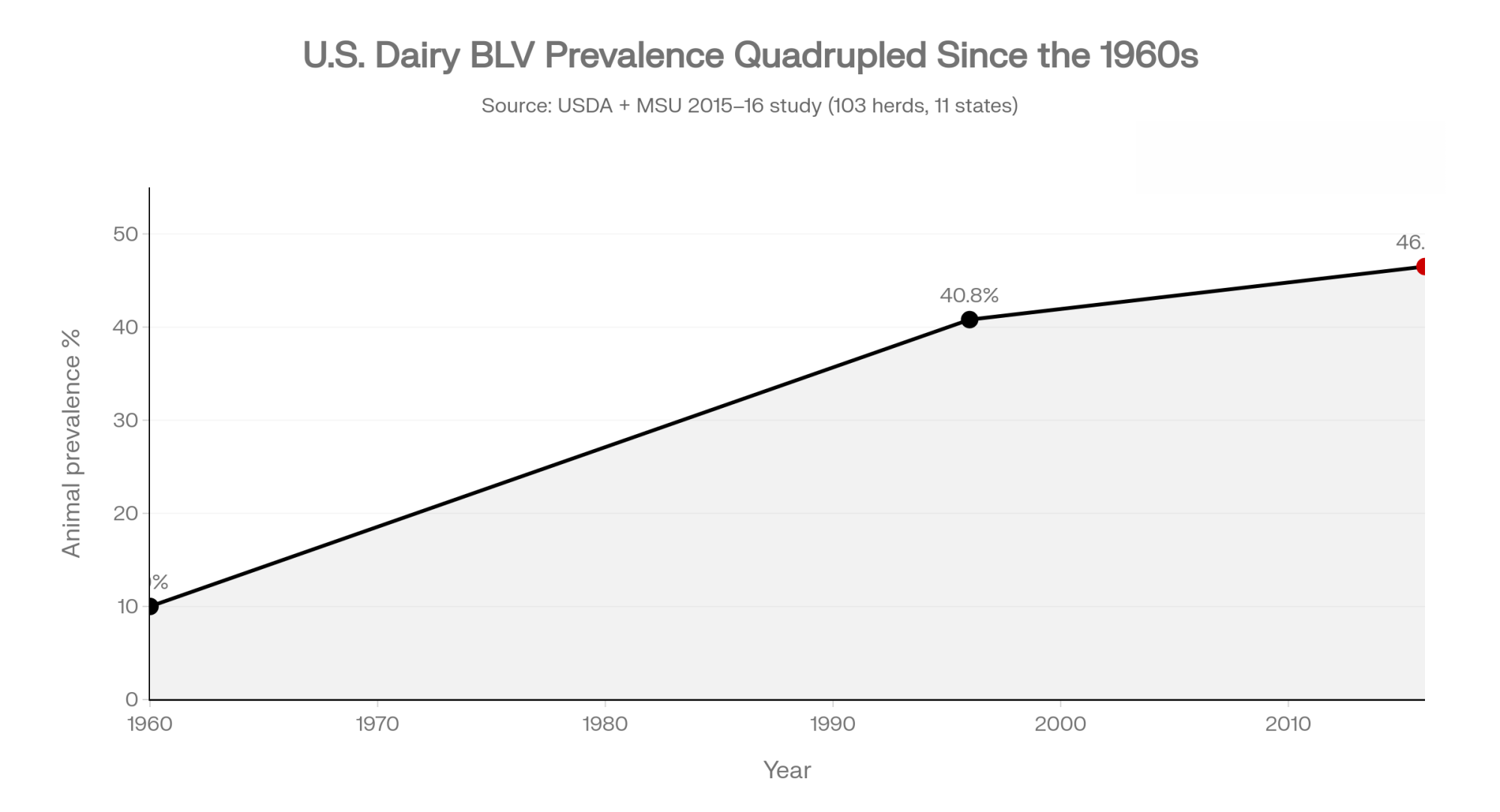
National work summarized by MSU’s BLV program shows 88.6–94.2% of U.S. dairy herds now have at least one BLV‑positive cow, with typical within‑herd prevalence in the 40–50% range. Animal‑level infection climbed from about 10% in the 1960s to 40.8% in a 1996 USDA study and around 46.5% in a 2015–2016 MSU national study of 103 herds across 11 states — the most recent national anchor point published by the team. In some high‑pressure regions, individual herds sit close to 100% positive.
Several European countries hit BLV early with coordinated test‑and‑cull programs and wiped it out while herd prevalence was still under 5%. The U.S., Canada, Japan, Argentina, and others largely did not, and the virus took the room it was given.
Kendrick, who works with the MSU BLV research team, put it plainly on The Dairy Health Blackbelt Podcast: infected cows “have lower milk production, decreased longevity” and a weaker immune system, even when they look normal in the pen.
Economic modeling using U.S. data from the mid‑1990s through the 2010s estimates that at around 50% prevalence, BLV costs roughly $6,400 per 100 milking cows per year in unrealized milk, shortened productive life, and lost salvage value. On a national scale, MSU reports BLV losses of about $525 million in 1996, with more recent figures suggesting an annual deficit of up to $2.7 billion.
You’ll never see “BLV” on your milk cheque. You feel it as 200–300 lb less milk from certain cows, more mastitis and respiratory cases that never quite clear, older cows dying instead of shipping, and condemnation slips that turn a lifetime of investment into zero salvage at the plant.
What BLV Is Quietly Doing Inside Your Cows
BLV is a deltaretrovirus, closely related to human T‑cell leukemia virus, that targets B‑lymphocytes — especially those expressing CD5 and CD21 markers. It integrates its DNA (the provirus) into those cells and turns them into long‑term virus factories. Your cows’ immune systems still work, but they work with a constant handicap.
Researchers and field programs see three big knock‑on effects:
- Weaker vaccine response. BLV‑infected cows often mount a reduced antibody response, so the vaccines you buy don’t deliver full protection.
- Higher disease burden. Positive animals show higher rates of mastitis, respiratory issues, and lameness, especially as they get older.
- Chronic inflammation. Studies in BLV‑positive heifers, particularly during transition, have found elevated haptoglobin and fibrinogen — markers of ongoing inflammation that quietly drain energy and resilience.
Clinically, about 70% of infected cows are aleukemic carriers with no obvious signs, around 30% develop persistent lymphocytosis, and fewer than 5% go on to full enzootic bovine leukosis with visible tumors. From a business point of view, that 70% matters more than the 5%, because those “normal‑looking” cows still cost you milk and lifetime production.

MSU work led by Bartlett, Erskine and colleagues links every 10‑point increase in BLV prevalence to about 209 lb less milk per cow per year, with a corresponding hit to the rolling herd average. A follow‑up MSU survival analysis tracking 3,849 animals across 112 herds for roughly 19 months found ELISA‑positive cows were about 23% more likely to leave the herd through culling or death, with the effect growing as antibody levels rose (Bartlett et al., Journal of Dairy Science 96:1591–1597, 2013). BLV‑related lymphoma is now the number one reason for U.S. dairy cow carcass condemnation, accounting for 26.9% of dairy condemnations and 13.5% in beef cows, per USDA FSIS data summarized in the MSU materials.
Worst of all, those extra deaths and condemnations cluster in older cows, right when they should be printing margin — peak lactations plus a final salvage cheque. BLV quietly “prunes” those cows out of your herd early. Less lifetime milk. Less salvage. Less room for mistakes elsewhere.
What a BLV Herd Looks Like on Paper
The typical pattern MSU and other BLV teams describe in 40–50% prevalence herds is a year of records that never quite add up — nothing that screams BLV, but a persistent background of repeat mastitis cases, hard‑to‑resolve respiratory episodes, and non‑calving deaths in 3rd‑plus lactation cows.

Per 100 milking cows, a herd at that prevalence often shows a pattern like this in the records (illustrative numbers stitched from MSU research and field observations, not a single farm’s dataset): around 32 clinical mastitis cases, with a handful of older cows making repeat appearances; roughly 9 respiratory or “fever of unknown origin” episodes; about 14 lameness events outside the normal trim schedule; and just under 6.5 non‑calving deaths or euthanasias, many in 3rd‑plus lactation cows. Rolling herd average? Respectable. BLV? Never discussed at the monthly meeting.
Find Your Herd on the BLV Tax Table
Using MSU’s modeled $283–$380 per infected cow per year at a typical 45% within‑herd prevalence, estimated annual losses scale with herd size. Actual losses on a given operation will vary with milk price, cull value, and management:
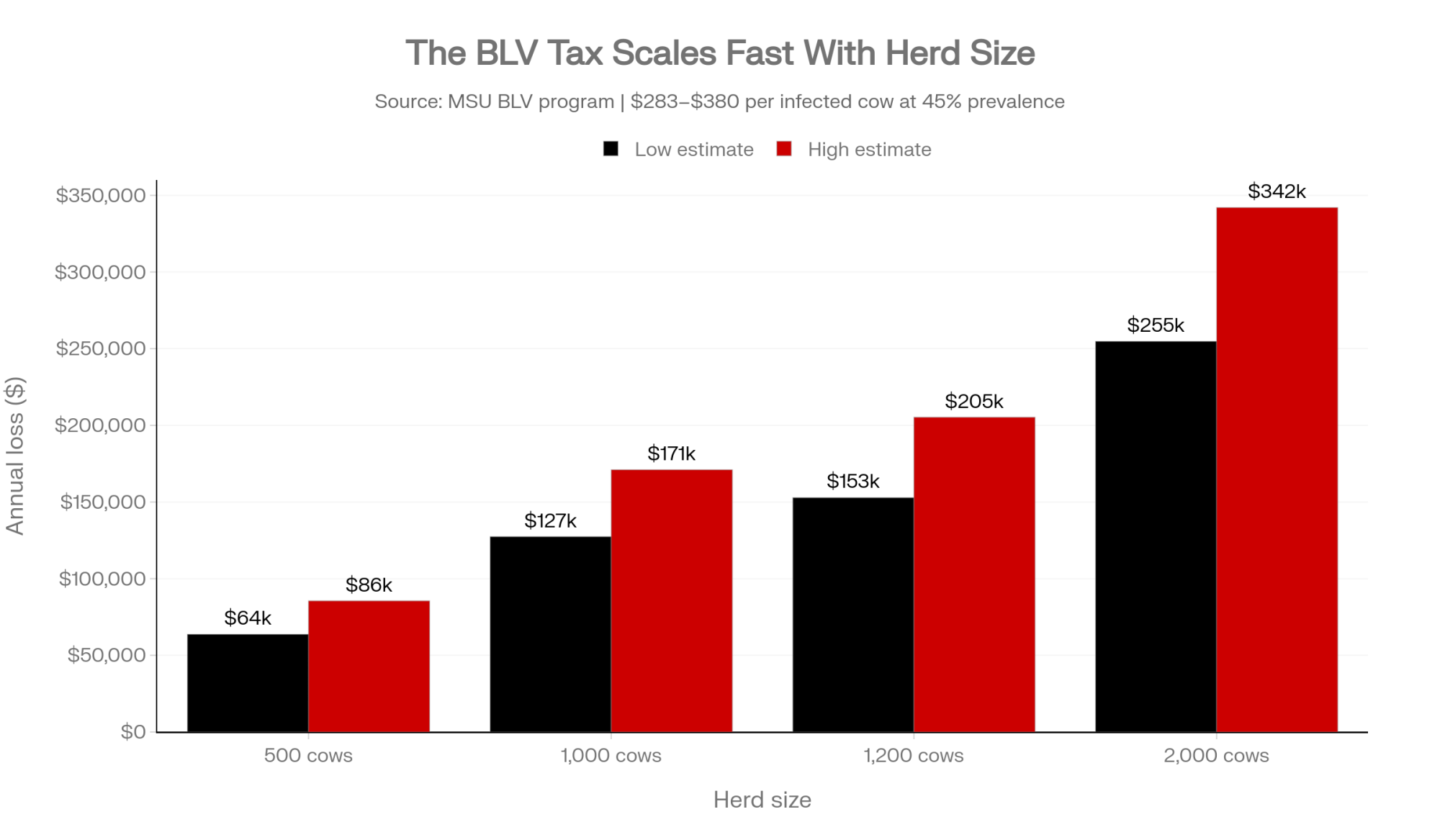
| Herd Size | Prevalence | Infected Cows | Annual “BLV Tax” (Est.) |
| 500 cows | 45% | 225 | $63,675 – $85,500 |
| 1,000 cows | 45% | 450 | $127,350 – $171,000 |
| 1,200 cows | 45% | 540 | $152,820 – $205,200 |
| 2,000 cows | 45% | 900 | $254,700 – $342,000 |
Once those numbers hit the kitchen table, BLV stops being a “we should look at that someday” topic and becomes a six‑figure line item you either accept or start clawing back.
The 4 A.M. Reality: Where Your SOPs Are Failing
If you’re going to fight something, you need to know how it travels. BLV moves with infected lymphocytes — mostly in blood, but also in colostrum and milk. On the ground, that turns into a few familiar trouble spots.
MSU’s BLV program and Dr. Kendrick’s work point to these high‑risk routes:
- Needles and syringes. Reusing needles, or leaving one “barn needle” stuck in a multi‑dose bottle all day, is a very efficient way to share infected blood.
- Palpation sleeves. Even when a rectal sleeve looks clean, micro‑tears in the rectal mucosa can carry blood from one cow to the next.
- Surgical and hoof‑trimming tools. Gouge‑type dehorners, tattoo pliers, and hoof‑trimming equipment that aren’t disinfected between animals become mechanical BLV buses.
- Biting flies. Flies act like dirty syringes, picking up infected blood on their mouthparts and injecting it into the next cow they bite.
Vertical and calf routes matter as well. BLV can cross the placenta in roughly 4–18% of pregnancies. More damaging for your future herd is what happens in the calf barn: in some herds, about 12% of young stock are already BLV‑positive before they ever enter the parlor, mainly through raw colostrum or waste milk from infected dams. If BLV‑positive milk is feeding your replacements, you’re essentially seeding your future herd with the virus.
The good news: colostrum is one area where you can win big with straightforward management. Pasteurizing colostrum at 60°C for 60 minutes or freezing and thawing it correctly will inactivate BLV while preserving the antibodies calves need. That’s one of the fastest ways to stop loading new animals into the BLV column.
Go deeper: the colostrum protocols that actually protect replacements — our Tier 2 calf‑management pillar.
How Much Is Your Herd Really Paying to Wait?
If you haven’t tested for BLV in years — or ever — it’s easy to assume your herd is “probably average.” Michigan State’s 40‑cow protocol exists to replace that guess with an actual number.
Here’s the method:
- Test 40 cows — 10 in each of four lactation groups: 1st, 2nd, 3rd, and 4th‑plus.
- Choose the most recent fresh cows in each group, making sure they’re at least 3 days in milk.
- Don’t cherry‑pick “good” or “bad” animals — take them in order.
Once you know that prevalence number, your BLV tax math is two steps:
- Milking cows × prevalence = number of infected cows.
- Infected cows × $283–$380 ≈ annual BLV tax range.
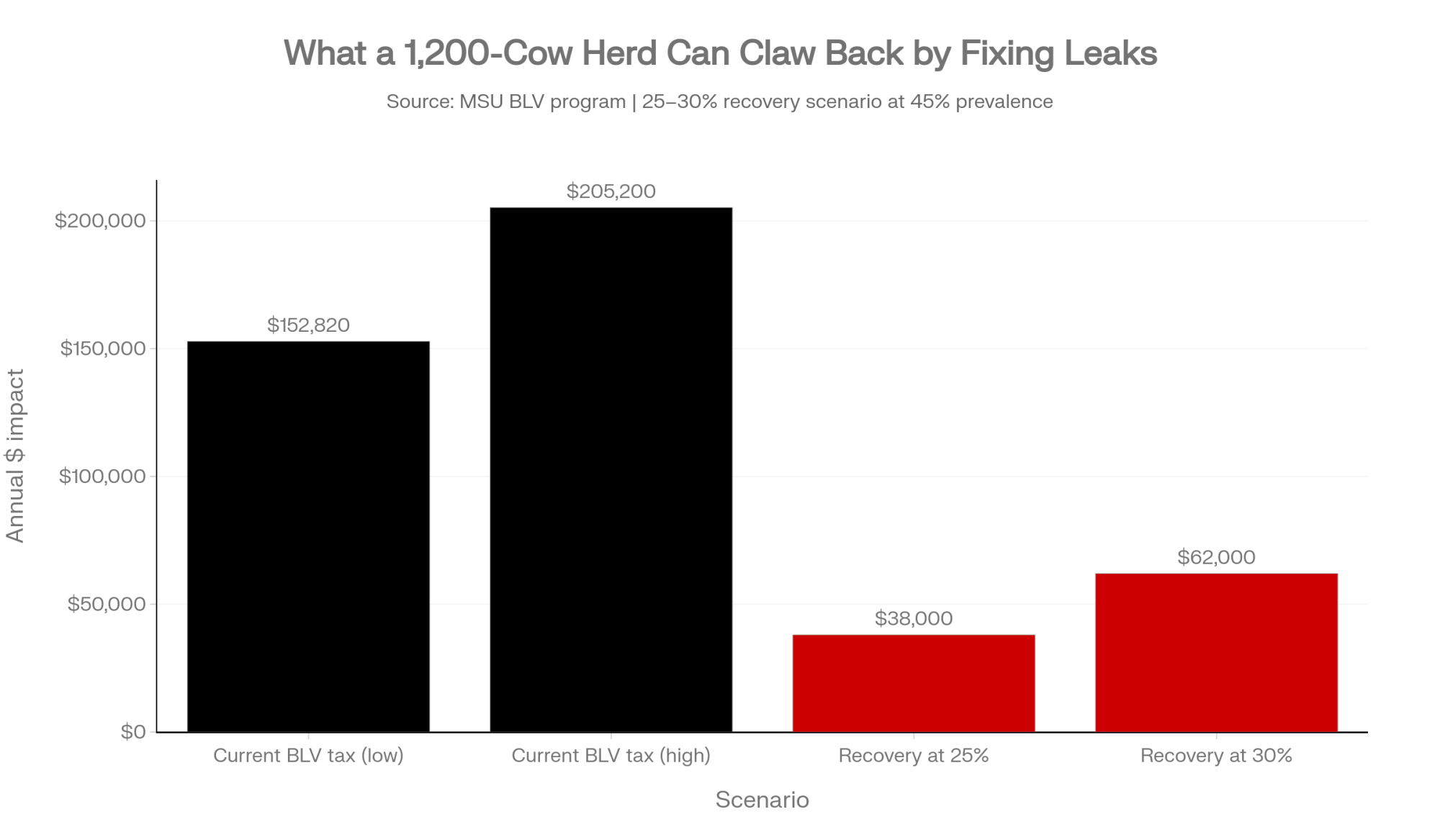
On a 1,200‑cow herd at 45% prevalence, that formula gives 540 infected cows and the $152,820–$205,200 range in the table above. It’s not perfect to the dollar, but it’s good enough to answer two questions. First, are you willing to keep paying that BLV tax? Second, if not, how much of it do you realistically want back over the next few years?
Even a conservative 25–30% recovery — by tightening the obvious leaks and dealing with the highest‑risk cows — can put roughly $38,000–$62,000 a year back into a 1,200‑cow herd at that prevalence. That’s not lottery money. It’s the difference between replacing equipment on schedule and nursing it through one more breakdown season.
The Super‑Spreader Problem: Why 11% of Cows Drive 64% of New Infections
Once you’ve seen your number, the uncomfortable part is looking in the mirror. Where is BLV still hitching rides in your routines? When herds run the 40‑cow test, make changes, then re‑test 12–18 months later, the BLV teams keep finding the same pattern. BLV prevalence drops a little, then stalls. When you dig into why, the answers usually fall into three buckets.

1. The “paper SOP vs. 4 a.m. reality” gap. BLV researchers describe a common pattern across many farms: a single needle left in a multi‑dose bottle for half the day, or a used syringe grabbed in a rush when someone is catching up. The same thing can happen in the repro line when a sleeve gets rinsed and reused “just this once.” Your protocol binder may say “new needle for every cow,” but those shortcuts are exactly the gaps BLV uses to move.
2. Super‑spreaders still living in high‑traffic pens. BLV doesn’t spread evenly across positives. In one 199‑cow herd summarized in MSU’s research, 11% of ELISA‑positive cows were responsible for about 64% of new infections. In another herd, just two cows carried 26% of the total proviral load (PVL). If those highest‑PVL cows are still in the fresh group, still donating colostrum, and still getting “one more chance” when they crash, you’re trying to drain the pool while the fire hose is still wide open.
Pro‑Tip: ELISA tells you who’s infected. qPCR tells you who’s dangerous. A standard ELISA test gives you a yes/no on exposure — good for herd‑level prevalence, but it treats every positive cow the same. Quantitative PCR (qPCR) goes further: it measures each cow’s proviral load (PVL) and reports a cycle threshold (Ct) value. The lower the Ct, the more virus in that cow’s blood, and the more likely she’s a super‑spreader. That’s how you move from “half my herd is positive” (which feels hopeless) to “these 10–15% of cows are driving most of my new infections” (which is a management plan).
3. A calf program that keeps re‑loading the virus. If your heifers are getting raw colostrum or waste milk from BLV‑positive or unknown cows, that ≈12% infected youngstock figure from MSU research is probably happening in your pipeline. The fix that works on real farms is blunt but manageable: colour‑code colostrum jugs (green for BLV‑negative, yellow for cows whose colostrum must be pasteurized, red for high‑PVL or unknown cows whose colostrum never goes to calves), keep a dedicated “calf‑approved” fridge, and make it a hard rule that calf feeders don’t grab anything else.

| Dimension | ELISA (antibody test) | qPCR (proviral load test) |
| What it measures | Antibody presence (yes/no exposure) | Quantitative virus copies per cell (Ct value) |
| Best use case | Herd-level prevalence baseline, MSU 40-cow profile | Identifying the top 10–15% super-spreaders |
| Resolution on risk | Treats every positive cow the same | Separates low-risk carriers from high-PVL spreaders |
| Management output | “Half my herd is positive” (feels hopeless) | “These 10–15% of cows drive most new infections” |
| Typical cost tier | Lower per sample | Higher per sample, targeted subset |
| Action it unlocks | Prevalence tracking, trend monitoring | Terminal-cull list, colostrum colour-coding, beef-semen assignment |
None of that’s glamorous. It’s sleeves, needles, tools, flies, and colostrum. But that’s where BLV actually moves — and where you can actually stop it.
Go deeper: why your best cows keep leaving too early — a closer look at BLV’s quiet effect on longevity.
Options and Trade‑Offs for Farmers
30‑Day Action: Run the 40‑Cow Test and Audit the Obvious
When it makes sense: You haven’t profiled BLV in the last few years, or have never tied BLV status to real dollars.
Your 30‑day checklist:
- ✅ Book the MSU 40‑cow BLV profile through your vet or lab.
- ✅ Pull the right cows: 10 most recent fresh animals in each lactation group, all at least 3 days in milk.
- ✅ Spend one week spot‑checking how injections, preg checks, dehorning, hoof trimming, fly control, and colostrum actually happen — not how they’re written down.
- ✅ Walk your crew through the BLV Tax Table using your own herd size and a realistic prevalence range.
Risks and limits: You may not like the prevalence number. You may also find your real‑world routines don’t match your SOPs. But staying blind is usually more expensive than finding out.
90‑Day Action: Protect Calves and Tag the Super‑Spreaders
When it makes sense: Your 40‑cow profile comes back at or above roughly 30–40% prevalence, and you expect to be milking in five years.
What it requires: Put a colour‑coded colostrum system in place and commit to pasteurizing or freezing any colostrum from BLV‑positive or unknown cows before it goes to heifers. Work with your vet to run qPCR on a subset of ELISA‑positive cows — usually older, high‑antibody animals — to identify the highest‑PVL individuals. Mark those cows in your herd software and in the parlor as terminal BLV cows: they get beef semen only, their colostrum never goes to replacements, you don’t spend on heroics when they crash, and they go to the top of the cull list as soon as they’re saleable.
Risks and limits: You’ll discard some colostrum and lean harder on frozen stores. You may ship a few older, good‑milking cows earlier than you’d prefer. You’re trading a bit of short‑term milk from a small group for lower infection pressure and healthier replacements over the long term.
Forward‑looking signal: As research into BoLA‑DRB3 alleles and BLV resilience matures, expect to see sires marketed not just for components or fertility but for keeping daughters at lower PVL even when infected. That’ll be another lever — but it won’t replace sleeves, needles, and colostrum work.
365‑Day Check: Re‑Test, Re‑Score, and Decide How Hard to Push
When it makes sense: You’ve made changes and want proof they’re working.
| Horizon | Trigger to act | Core action | Required investment | Cost of skipping |
| 30 days | Haven’t profiled BLV in years, or never tied status to dollars | MSU 40-cow ELISA profile + one-week SOP audit (needles, sleeves, colostrum, flies) | Lab fees for 40 tests; 1 week of walkaround time | Continuing to pay $152,820–$205,200/yr blind on a 1,200-cow herd |
| 90 days | Profile returns ≥30–40% prevalence | Colour-coded colostrum system + qPCR on older high-antibody cows + terminal-cull list for top 10–15% PVL | Pasteurizer/freezer capacity; qPCR subset; beef semen on flagged cows | ~12% of heifers entering parlor already BLV-positive via raw colostrum |
| 365 days | 12 months post-intervention | Re-run 40-cow profile, compare hospital pen, non-calving deaths, drug spend, cull age | Second round of ELISA tests; vet/nutritionist/bookkeeper review meeting | No evidence trail — can’t defend the investment or tune the plan |
| Ongoing | Prevalence stuck at ~same level after 12 months | Diagnose the leak: super-spreaders in fresh pens, slipped single-use needle rule, red-tag colostrum reaching calf fridge | Management attention, not capital | 2–3 point drift feels like “progress” while BLV tax keeps compounding |
What it requires: Re‑run the MSU 40‑cow BLV profile 12 months after your first test, using the same selection rules. Sit down with your vet, nutritionist, and bookkeeper and compare BLV prevalence then vs now, mastitis and respiratory cases per 100 cows, non‑calving deaths, drug spend, and cull patterns — especially in older cows. If prevalence has moved down meaningfully — say, in the range of 5–10 points, alongside a cleaner hospital pen and fewer old‑cow deaths — you have real evidence you’re clawing back part of the BLV tax.
Risks and limits: If prevalence only moves 2–3 points, it’s not a failure — it’s feedback. It’s your cue to look harder at where BLV is still moving: are super‑spreaders still parked in fresh pens, did single‑use needles quietly slide back, are red‑tag colostrum jugs sneaking into the calf fridge at 2 a.m.? BLV control is a multi‑year project, not a one‑and‑done shot.

Key Takeaways
- If your 40‑cow profile comes back at or above roughly 30–40% BLV prevalence, treat BLV as a structural cost on your P&L, not just a lab result to file away.
- If you can’t honestly say every cow gets a new needle and a new rectal sleeve every time, assume BLV is still catching rides and fix that before you spend big on extra testing or culling.
- If your non‑calving deaths and chronic mastitis cases cluster in older BLV‑positive cows, talk with your vet about high‑PVL qPCR testing and a terminal culling rule for roughly the worst 10–15% of positives — the small group most likely to behave like super‑spreaders in your herd.
- If raw colostrum or waste milk from BLV‑positive or unknown cows is feeding your heifers, treat that as your fastest leak to plug: move to pasteurized or frozen colostrum from BLV‑negative or low‑risk cows and keep “red‑flag” jugs out of the calf room.
- If you decide to invest in BLV control, put a 12‑month re‑test date on the calendar now and commit to measuring change in black and white instead of guessing.
You already know the “mystery” cows in your barn — the ones that eat up time in the hospital pen, come back on the list a month later, or die right when you expect one last lactation and a salvage cheque. The hidden question is whether BLV is quietly pulling more of those strings than you’d like to admit.
If you ran your own herd through the BLV Tax Table above, where would your number land — $63,000, $150,000, $300,000‑plus a year? And if you saw that figure in black and white, what would it take for you to cut a third of it over the next three years?
In our next segment of The Silent Tax series, we’ll dive into the specific ROI of high‑PVL culling — exactly how fast a herd can expect to see a return on its testing investment, and how the cost curve shifts at 30%, 45%, and 60% prevalence. For now, the first move is simple: run the 40‑cow test, watch your own barn for a week, and decide how much longer you’re willing to pay the BLV tax without a plan.
Complete references and supporting documentation are available upon request by contacting the editorial team at editor@thebullvine.com.
Learn More
- Dam-Sourced Colostrum Drops Calf Mortality from 8.9% to 6.2% – Inside the 93-Herd Protocol — Arms you with the exact 93-herd playbook to eliminate pooled waste milk risks. Tightening teat-replacement schedules and guaranteeing dam-sourced first meals cuts mortality by 2.7%, shutting down a major viral entry point before replacements reach the parlor.
- 9.57 Million Cows, 3.9 Million Replacements: Genetics Built This Dairy Herd Paradox – and 2027 Ends It — Follows the money on the national heifer shortage to expose why forced retention crushes your bottom line. With replacements costing over $3,010, losing mature cows to early disease culls destroys margin far faster than typical herd turnover.
- The $3,500 Calf Question: What Dairy Farmers Need to Know About April 2026’s New CDCB Calf Health Evaluations — Breaks down the CDCB’s latest genomic release to help you breed resilience directly into your future herd. Moving beyond basic management protocols, selecting for these newly independent disease resistance traits pushes mortality toward an elite 1-2% range.
 The Sunday Read Dairy Professionals Don’t Skip.
The Sunday Read Dairy Professionals Don’t Skip.
Every week, thousands of producers, breeders, and industry insiders open Bullvine Weekly for genetics insights, market shifts, and profit strategies they won’t find anywhere else. One email. Five minutes. Smarter decisions all week.






 The Sunday Read Dairy Professionals Don’t Skip.
The Sunday Read Dairy Professionals Don’t Skip.