California dairy shows are back. Now every entry form is a three-way bet on genetics, H5N1 risk, and whether your kids’ hard work really pays.
Executive Summary: California dairy shows are back on the calendar after CDFA lifted its statewide H5N1 exhibition ban, allowing poultry and dairy cattle to return to fairs and junior rings in 2026. That call comes after a year where H5N1 touched more than 700 California dairy herds and about three‑quarters of all U.S. HPAI‑affected farms were inside one state—a level of risk that shut down barns, youth projects, and show‑driven marketing plans overnight. Now the pressure shifts back to you: every entry form in 2026 is a three‑way bet on genetics promotion, H5N1 exposure, and whether your kids’ hard work ends up paying off or just adding stress. This article breaks down how the ban unfolded, what it actually cost fairs, youth programs, and breeder herds, and why state officials finally felt confident enough to let cattle back onto the shavings. Most importantly, it gives you a clear, kitchen‑table playbook—how to talk through risk with your vet, decide whether you’re a show‑forward, selective, or stay‑home herd this year, and make sure any banner you chase still pencils out in terms of Net Merit, PTAT, components, and long‑term profit.

Here’s what’s really going on. After a full year of empty dairy barns and pulled classes, California has finally flipped the switch. The statewide ban on poultry and dairy cattle exhibitions at fairs and shows is gone, and the show community is rushing—some excited, some cautious—to figure out what 2026 actually looks like.
If you’re running a serious herd in California, or you sell genetics into that market, this isn’t just a nice headline. It changes how you market cattle this year, how you manage disease risk, and what your 4‑H and FFA kids are working toward when the barn lights come on at 5 a.m.
How We Got Here: From Full Stop to “You’re Back On”
In early January 2025, California’s State Veterinarian, Dr. Annette Jones, sent a letter that landed on fair boards across the state: all poultry and dairy cattle exhibitions at fairs and shows were banned, effective immediately and “until further notice.” That wasn’t an academic exercise in risk management—it was a response to a very real fire.
By that point, state and local reports showed that more than 700 dairy herds and around 60 poultry flocks in California had already been affected by H5N1, with over 15 million birds lost or depopulated. Hoard’s Dairyman’s “Washington Dairygrams” reported that, as of early January 2025, 918 farms in 16 states had confirmed HPAI, and 703 of them were in California. Roughly three‑quarters of every infected farm in the country was in one state.
That’s not a “we’ll keep an eye on it” moment. That’s a “we need to stop giving the virus free rides” moment.
The logic behind the exhibition ban was brutal but clear:
- Fairs and shows bring cattle and poultry from many herds into the same barns and wash racks.
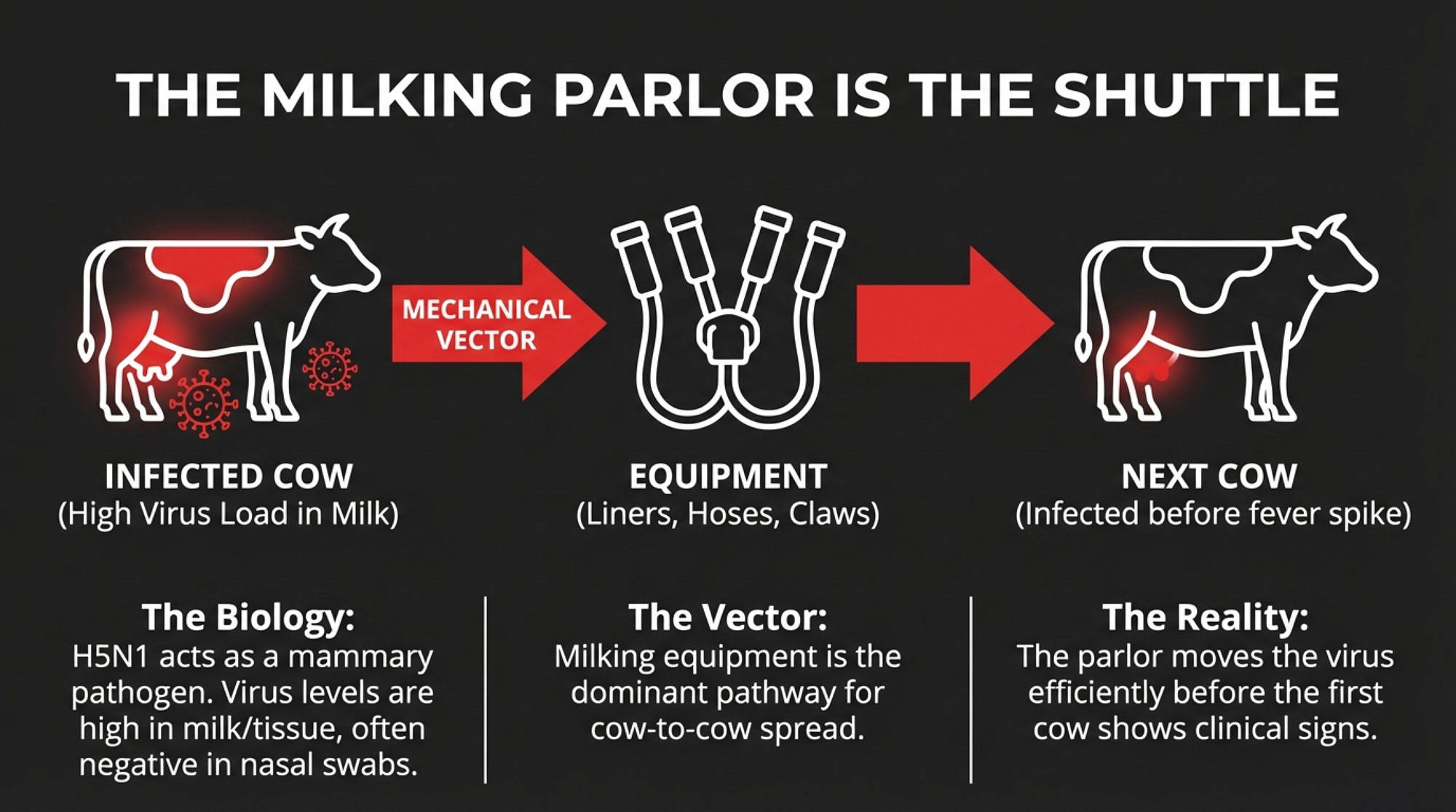
- Field investigations and early research showed infected dairy cows often had subtle signs—lethargy, drop in yield, off feed—rather than the dramatic mortality we see in poultry, which makes it easy to miss unless you’re testing and watching carefully.
- Genetic sequencing linked H5N1 viruses in some California dairy herds to nearly identical strains in Colorado, pointing straight at cattle movement and people, not just wild birds, as key drivers.
CDFA’s ban language said the goal was to “minimize the danger of exposing people and non‑infected cows and birds to the disease.” With 703 of 918 infected farms nationwide sitting inside California, there weren’t many soft options left.
So the state did the one thing it could control quickly: it shut down the highest‑risk mixing points—poultry and dairy cattle at fairs and shows.
On the other end of the curve, CDFA didn’t just let the order quietly expire. After months of surveillance, trend analysis, and consultation with state and federal epidemiologists, Dr. Jones issued a new letter in December 2025 announcing that the statewide ban on poultry and dairy cattle exhibitions was lifted, effective immediately. On January 16, 2026, CDFA followed with a public release confirming that fairs and shows could bring poultry and dairy cattle back, citing:
- Improved H5N1 infection trends.
- A better understanding of how the virus behaves in dairy cattle.
- Stronger mitigation and biosecurity tools at farm and fair levels.
So yes, the state has changed the rule. But the risk management has been handed back to you.
What the Ban Actually Cost: Youth, Barns, and Banners
Let’s talk about what this did where you live: in the barns, not in the press releases.
Youth projects ran into a wall
CDFA’s “Statewide Ban on Poultry and Dairy Cattle Exhibitions” FAQ doesn’t dance around who’s affected. It names 4‑H, FFA, Grange, and independent youth exhibitors as part of the ban and explains that dairy cattle in those programs cannot be shown while the order is in place. The ban specifically targeted the highest‑risk dairy classes: lactating and recently lactating cows and replacement heifers—the exact animals most dairy kids show. cdfa.ca
For a 13‑year‑old, losing one fair is disappointing. For a 17‑year‑old senior, it can be the difference between walking into the ring for a final year and aging out without that moment. UC ANR materials and county messaging describe county fairs as core agricultural education spaces; in 2025, dairy projects simply weren’t part of that picture.
We’re not naming specific kids in this article, and that’s on purpose. Until juniors and families are ready to tell their stories on the record, we’re not going to pretend we know exactly whose senior year got cut short. What’s clear from the record is that a lot of youth dairy work had nowhere to land last year.
County fairs lost more than a couple of classes
Take dairy and poultry out of a county fair, and the whole atmosphere shifts.
The California Mid‑State Fair publicly announced in March 2025 that all live poultry and dairy cattle exhibits were removed for that year because of “the alarming spread of bird flu across the state” and to comply with the State Veterinarian’s ban. San Joaquin’s AgFest and similar events posted the CDFA orders and told families outright that dairy cattle weren’t coming through the gates in 2025.
The Santa Cruz County Fair, which operated under that ban, is now making a point of discussing its return. In January 2026, CEO Dori Rose Inda said, “We are thrilled to have poultry and the dairy cows back with the amazing youth that care for them and show them at the 2026 Fair,” framing the animals and the kids as core to what makes the fair feel like a fair again.
You don’t have to be a show junkie to know that when the dairy barns go quiet, the rest of the grounds don’t feel quite right.
Show‑driven marketing plans stalled out
There’s another layer here. Shows aren’t just about trophies—they’re marketing infrastructure for genetics‑driven herds.
Operations like Vierra Dairy in Hilmar have built a national reputation by combining consistent show success—especially in Jerseys—with classification and genomic data to move embryos, live cattle, and bull contracts. Local and regional shows inside California have always been part of the pipeline that feeds into national stages and sales:
- Genomic heifers often get their first exposure there.
- Young cows get shown close to home to build momentum before a big sale.
- Junior members of key cow families get in front of local buyers and visiting breeders.
When California pulled dairy shows from fairs for 2025, that mid‑tier exposure vanished. We don’t have a precise dollar figure for lost entry fees, cancelled show consignments, or delayed sales—that’ll require fair data and breeder interviews in a follow‑up piece. But we do know entire segments of the show calendar simply didn’t happen.
If you rely on the show ring to help tell your genetics story, you lost a year of that storytelling.
The Herd‑Health Math: Why the Ban Actually Made Sense
It’s easy now to say, “They went too far.” Let’s walk through why, from a herd‑health perspective, the ban was the least bad option at the time.
The virus was moving with cattle, not just wild birds
Research and field reports from 2024–2025 pulled together a consistent picture:
- In dairy cattle, H5N1 often showed up as mild, non‑specific illness: reduced milk production, lower feed intake, and some respiratory signs—nothing like the catastrophic mortality seen in poultry barns.
- Genetic analysis linked H5N1 viruses in dairy herds in Texas, Colorado, and California; strains in some California herds were nearly identical to those in Colorado, strongly suggesting that cattle that fought the virus were moved between herds.
- USDA responded with a Federal Order requiring testing for Influenza A before lactating dairy cattle move across state lines and tightening reporting requirements for infected herds.
The big takeaway: H5N1 in dairy wasn’t just a wildlife problem. It was a movement and contact problem.
Now take that reality and look at a state fair dairy barn: cattle from different herds, shared equipment, shared wash racks, people bouncing from string to string, and no practical way to enforce strict separation without major structural changes.
From CDFA’s perspective, with 703 infected farms in California out of 918 nationally, those barns started to look like disease amplifiers rather than showcases.
By the time the ban hit, the fire was already burning
Could the state have tried intermediate steps—like “no lactating cows, heifers only,” or “negative tests for all show animals” first? Maybe, if they’d moved earlier. But the timeline matters: CDFA and fair accounts show H5N1 hitting California dairy herds and poultry flocks hard through late 2024, with the ban announced only after the numbers turned ugly.
By early January 2025, California wasn’t managing a handful of cases. It was carrying roughly three‑quarters of all HPAI‑affected farms in the country. At that point, shutting down high‑traffic exhibition points while they got a handle on transmission was the logical move, even if it landed hard on show families.
Where We Are Now: Risk Is Lower, But It’s Still Risk
So what changed enough for CDFA to say “yes” again?
In late 2025, CDFA, UC Davis extension, and local fair communications all point to the same factors when explaining the decision to lift the ban:
- Infection trends improved. Surveillance programs—milk testing, herd monitoring, and reporting—showed fewer new H5N1 detections in dairy herds, and known cases were being managed under quarantine and control programs.
- The industry understands the virus better. By then, USDA and researchers had months of data on how H5N1 behaved in cows, how long shedding lasted, and what worked to contain it in real dairy environments.
- Mitigation tools got sharper. Movement permits, testing requirements for certain categories of cattle, and farm‑level biosecurity improved compared to early 2024.
On that basis, CDFA lifted the statewide exhibition ban. But they’ve been very clear that lifting the ban is not an “all clear.” Current guidance to fairs and exhibitors emphasizes:
- Don’t haul or exhibit animals that look even slightly off.
- Use veterinary oversight aggressively at fairs and at home.
- Expect ongoing H5N1 monitoring and be ready for new guidelines if risk indicators change.
The risk environment is better, but you’re still operating with a virus in the background, not a virus that’s gone.
The First Real Chances to Get Back in the Ring
Let’s get specific about where your cattle and your kids might actually be headed.
County fairs are rebuilding dairy barns
California Mid‑State Fair – Paso Robles
The Mid‑State Fair’s 2025 decision to remove all poultry and dairy cattle exhibits is well‑documented. Now that the ban has been lifted, they can rebuild those programs for 2026. That means recruiting exhibitors back, updating health rules, and resetting expectations after a blank year.
Santa Cruz County Fair – Watsonville
Santa Cruz County Fair communications now highlight that poultry and dairy cattle will be back for the 2026 fair, with CEO Dori Rose Inda publicly celebrating the return of both the animals and the youth who care for them.
Other county fairs and agfests
San Joaquin AgFest and others have circulated CDFA’s FAQs and updates, encouraging exhibitors to stay on top of state guidance as they prepare to reintroduce dairy cattle and poultry. For most fair managers, 2026 looks like a rebuild year: same barns, new rules.
Western Classic and other dairy‑focused events
The Western Classic Junior Dairy Show in Tulare has long been a magnet for junior exhibitors and top‑end showstrings from across the West. Its published entry info and social channels now reflect the reality that California dairy shows are allowed again, making Western Classic one of the earliest big tests of how junior dairy shows function under the post‑ban biosecurity expectations.
On the breed side, California is still home to some of the most aggressive show herds in North America. Herds like Vierra have made a business case out of combining show results with proof sheets and classification to drive semen, embryo, and live‑animal demand. With California shows back on the calendar, those herds can once again use a local and regional ladder to support their national ambitions, instead of having to jump straight to out‑of‑state events.
The Hard Question: Is Hauling a String Worth It in 2026?
Here’s where it gets uncomfortable. Just because the ban is gone doesn’t mean hauling a showstring is automatically the smart move for your operation this year.
You’re balancing two real forces:
- The upside: genetics marketing, ROI on show cattle, youth development, and community connection.
- The downside: H5N1 and other disease risk from mixing cattle in shared barns, plus the financial and labor cost of showing.
What you stand to gain
If your operation leans on shows as part of your marketing engine, a functioning show season gives you tools you don’t get by just posting pictures on Facebook.
- A class‑winner or champion can bump:
- Embryo values for that cow family.
- Live‑animal sale prices on daughters and maternal sisters.
- Bull‑dam visibility when AI studs are sorting cow families and photos.
- Regional show results, tied to classification and genomic proofs, can give your online sales and consignment offerings credibility that pure numbers sometimes can’t.
Herds like Vierra have shown that when you line up banners, solid type, production, and genomic profiles, you can turn show success into real contracts and long‑term demand. That’s genetics ROI, not just bragging rights.
And for youth? Having a real show season again is about more than hardware. It’s about:
- Learning to manage animals under pressure.
- Dealing with judges, buyers, and the random person in the stands who doesn’t know which end of the cow is which.
- Figuring out if they actually see a future for themselves in agriculture.
Those are hard things to replicate without a show ring.
What you’re putting at risk
On the other side, the disease risk and cost structure haven’t magically disappeared.
- H5N1 is still present in North American dairy cattle; the difference now is more monitoring and better tools, not zero risk.
- Movement of cattle and people has been clearly implicated in the spread between herds and states.
- Fairs and shows remain high‑traffic environments for both animals and humans.
Layer on top of that:
- Entry fees.
- Hauling and fitting.
- Labor pulled off the home farm.
- The risk is that a problem in the string becomes a problem in the main herd if you don’t have good separation.
The smart question isn’t “Can we go?” It’s “For this show, and this animal, in this year, does the upside actually justify the cost and risk?”
A Practical 2026 Show Decision Checklist
Here’s a straightforward way to think this through—no fluff, just the questions you’d ask yourself around the kitchen table.
1. Get clear on why you’re showing
Before you send an entry, ask:
- Are we promoting a specific cow family ahead of a sale or a flush program?
- Are we trying to build a bull‑dam’s profile for AI conversations?
- Are we primarily doing this for youth development and family experience?
If the honest answer is “because we always go,” then you’re in the tradition/hobby lane. That’s okay if you can afford it—but don’t pretend it’s a pure business decision.
2. Be brutally honest about your herd health and physical setup
With your herd vet, walk through:
- Any unexplained production dips or health quirks in the last few months.
- How realistically you can separate show animals from the main herd:
- Separate pens or a dedicated show facility.
- Dedicated boots, coveralls, halters, pitchforks, and brushes for show cattle.
- A plan for how long returnees stay isolated and how you’ll watch them.
If separation is mostly theoretical, then any show‑related disease exposure is a herd‑level risk, not just a show‑string risk.
3. Put show organizers on the hook—in a respectful way
Before you invest:
- Ask if the show is working under the current CDFA guidance for poultry and dairy cattle exhibitions.
- Ask whether a veterinarian will be on-site and how they’ll handle an animal that appears ill in the barn.
- Ask whether any testing or additional health documentation is required this year.
If answers are vague, or you get the sense that “business as usual” is the only plan, that should factor into your decision.
4. Do a back‑of‑the‑envelope ROI on one key animal
For one heifer or cow you’re thinking of hauling, jot down:
- Her realistic value if she stays home this year.
- A realistic bump in value if she wins or places well—embryos, live sales, or bull contracts, not fantasy numbers.
- Total costs: entries, hauling, fitting, extra feed, plus your time.
- A rough “risk cost” in your head: what happens if she or a herd mate picks something up as a result of that trip.
If the upside is only marginally better than the combined cost and risk, you’re probably in hobby territory, not high‑ROI marketing. That may still be fine—but you should know which one it is.
Youth Exhibitors: They Lost More Than a Banner
For adults, a lost show season is a business and logistics problem. For youth, it can be the moment that either pushes them away from agriculture or pulls them deeper in.
CDFA’s FAQ and UC ANR summary both state that youth exhibitors were directly affected by the ban, and county fair leadership has been blunt that losing dairy and poultry removed a big piece of youth programming in 2025. Fairs like Santa Cruz are now emphasizing that bringing dairy cattle and poultry back is partly about restoring those youth opportunities, not just filling barns.
If you’re a parent, project leader, or advisor, your 2026 show choices send signals:
- That herd health and biosecurity matter in your operation.
- That you’re willing to weigh risk and reward out loud, instead of just “hoping for the best.”
- That you still see a future in dairy worth your kids’ time, even after a tough year.
The “right” answer—haul or don’t haul—won’t be identical for every family. But involving your kids in that reasoning process is part of what makes this more than just a season of banners.
What You Should Actually Do in the Next 60–120 Days
Let’s put this into a simple playbook you can actually use.
Step 1: Build a realistic 2026 show calendar
Contact the fairs and shows that matter to you and confirm:
- Dates and locations.
- Which dairy breeds and classes they’re offering in 2026.
- Any new health, testing, or biosecurity requirements for entries.
From there, build a calendar that fits your labor, cash flow, and animal inventory—not just the “we always go” list.
Step 2: Decide which “show herd” you are this year
Be clear which lane you’re in:
- Show‑forward herd: You’ve got the people, facilities, and genetics to target a handful of major shows. You invest in biosecurity and expect a marketing return.
- Selective marketing herd: You choose one or two strategic shows, then build photography, video, and sales messaging around those results.
- Stay‑home herd: You decide the H5N1 and logistics risk isn’t worth it this year. You lean into genomic proofs, classification, production records, SCC, components, and good storytelling to market cattle without leaving the yard.
All three strategies can work. The key is not lying to yourself about which you’re actually following.
Step 3: Build a show‑specific biosecurity plan with your vet
This year isn’t the year to wing it.
Ask your herd vet to help you set:
- Pre‑show health check criteria and any recommended testing.
- A clear isolation and monitoring protocol for animals returning from each event.
- Rules for people and equipment moving between the showstring and main herd.
Tie that plan directly to what USDA and CDFA have said about H5N1 in dairy cattle—subtle clinical signs, movement‑driven spread, and the continued need for surveillance.
Step 4: Make sure the show results feed your bottom line, not just your ego
If you’re investing in a show season:
- Make sure pedigrees, sire stacks, classification scores, and production records are ready to share alongside ring photos.
- Capture high‑quality pictures and video at shows and at home.
- Decide in advance how the show results will plug into:
- A sale catalog,
- An online offering,
- AI stud conversations, or
- A flush or IVF program.
Shows can still be a powerful lever. In 2026, that lever has to connect to Net Merit, PTAT, components, and long‑term herd strategy—not just a banner in the office.
The Bottom Line: The Ban Is Gone, but the Responsibility Isn’t
California lifting the exhibition ban tells you three things at once:
- The H5N1 situation is better than it was, but it’s still on the radar.
- The state now expects producers, vets, and fair organizers to do more of the practical risk management.
- The show ring is once again a legitimate tool for youth development and genetic marketing—if you use it intentionally.
The barns are open again. Whether that’s a net win for your operation depends on the choices you make from here—about herd health, about where you chase banners, and about the example you set for the next generation watching you call the shots.
Use shows where they genuinely support your herd, your kids, and your business—not just because “we always go.” Respect the virus, respect your neighbors, and don’t assume another emergency order is impossible.
Everyone would like this reopening to stick. Whether it does will come down to how carefully fairs, exhibitors, and vets handle the next season.
We Want Your Story
If you’re a California exhibitor—youth or adult—who lived through this ban, we’d like to hear your side:
- What year of your project did you lose?
- Which cow or heifer never got her shot in the ring?
- What sale or marketing plan did you have to shelve?
- Where will you take your string first now that the barns are open?
Email andrew@thebullvine.com or message The Bullvine on Facebook. With your permission, we’ll build a follow‑up feature around the people behind these decisions—because policies and case counts matter, but it’s your lives, your cows, and your communities that make this story worth telling.
Key Takeaways
- Shows are back—risk isn’t: CDFA lifted California’s year-long H5N1 exhibition ban, but the virus is still out there. Every entry form in 2026 is a three-way bet on genetics, disease exposure, and whether the trip actually pays.
- Youth projects took the hardest hit: 4-H, FFA, and Grange kids sat out an entire season. For seniors, that wasn’t a setback—it was their last ring, gone.
- Show-driven marketing lost a year: Herds that use banners to move embryos, build bull-dam profiles, and drive sale prices had no California stage in 2025.
- Now you run the numbers: Decide if you’re show-forward, selective, or staying home—and tie every show decision back to Net Merit, PTAT, components, and long-term herd profit, not nostalgia.
- Biosecurity is the price of entry: Work with your vet on pre-show health checks, return isolation protocols, and strict separation between showstring and main herd. Winging it isn’t a plan anymore.
 Join the Revolution!
Join the Revolution!
Join over 30,000 successful dairy professionals who rely on Bullvine Weekly for their competitive edge. Delivered directly to your inbox each week, our exclusive industry insights help you make smarter decisions while saving precious hours every week. Never miss critical updates on milk production trends, breakthrough technologies, and profit-boosting strategies that top producers are already implementing. Subscribe now to transform your dairy operation’s efficiency and profitability—your future success is just one click away.






 Join the Revolution!
Join the Revolution!